Prof. Artur Baszko: Ablation procedures in congenital heart diseases
Ablation procedures in congenital heart diseases are a challenge not only for the complexity of anatomical substrate, but also due to the limited access to the heart. A vascular access for the electrophysiological study (EPS) is most commonly obtained via the femoral vein. Therefore transseptal catheters and electrodes are adapted to this access type. However, placement of electrodes into the coronary sinus is feasible using either the femoral access or an access achieved by puncture of jugular or subclavian vein.
Inferior vena cava (IVC) obstruction may be congenital (occurs in approximately 0.15% of general population and approximately 0.6% of patients with congenital heart diseases) or acquired. The acquired obstruction may be caused by previous surgical procedures, cardiac catheterization, thrombosis or trauma.
Publications on electrophysiological procedures in patients with IVC obstruction have been reviewed in a recently published paper. The authors found that the obstruction was congenital in the majority (80%) of 142 analyzed patients. Main procedures performed on this population were: pulmonary vein isolation, atrioventricular nodal reentry tachycardia and atrial flutter ablation. A focal atrial tachycardia (AT) was diagnosed in 9 patients, while macro-reentrant AT was diagnosed in 11 patients.
Yet, there is still a limited data concerning the management of postoperative AT in pediatric patients with obstruction of both femoral or iliac arteries found during the EPS. In our experience, a Doppler ultrasound performed before EPS frequently showed patency of the venous system, while veins were in fact obstructed on higher levels. It is also known that the use of multipolar electrodes for high-resolution mapping significantly improves the efficacy of mapping and ablation in those patients. In available literature we found no paper describing experiences with the use of the HD Grid Advisor electrode in the case of IVC obstruction.
Here, we present a case of 14-year-old child with recurrent drug-resistant AT that underwent atrioventricular septal defect repair (2007) and mitral valve repair with Bioring (2010), and with moderate mitral stenosis. Cardiac catheterization performed in this patient led to diagnosis of irreversible pulmonary hypertension. Due to sinoatrial node dysfunction the child had a VVI stimulator with epicardial electrode implanted (2010). Since the beginning of 2011 the patient underwent electrical cardioversion nearly every month for unstable atrial tachycardia. The tachycardia led to presyncope related to fast conduction to the ventricles (at rate of 190-210/min). In periods between arrythmia recurrences ventricular rhythm with retrograde conduction to the atria was dominating.
The ablation procedure and upgrade to dual-chamber pacemaker, that could restore atrioventricular synchrony, were considered as potential therapeutic options. It was decided, however, to focus on the ablation and to postpone the decision on pacemaker mode modification from ventricular to dual-chamber.
Before the ablation procedure, a Doppler ultrasound of the venous system was performed. No significant femoral veins anomaly was detected.
The ablation procedure was performed under general anesthesia achieved with the use of fentanyl, propofol and sevoflurane.
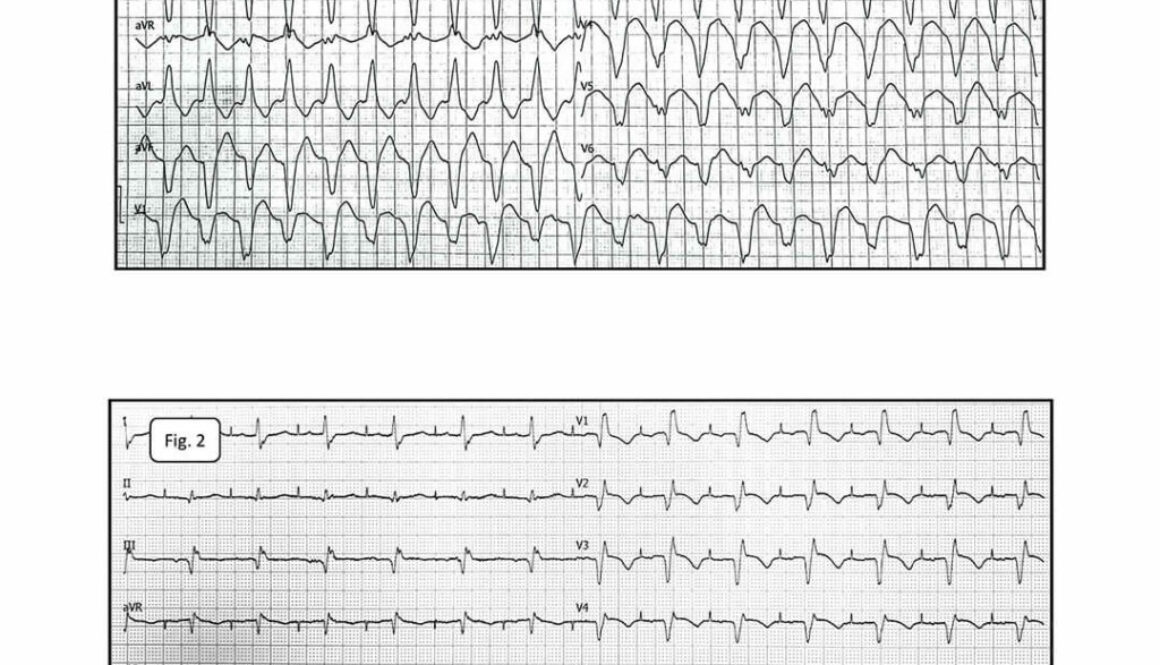
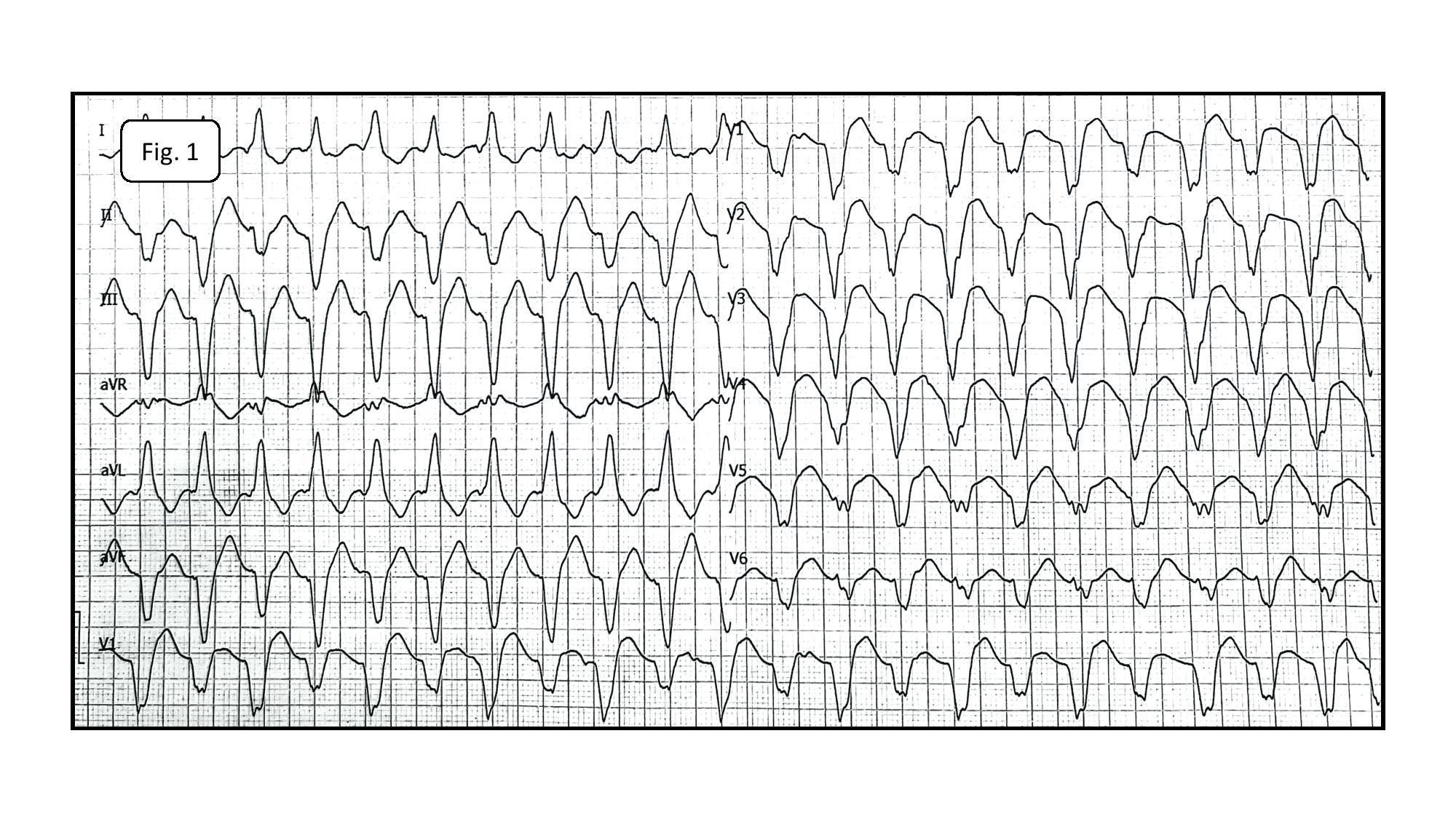
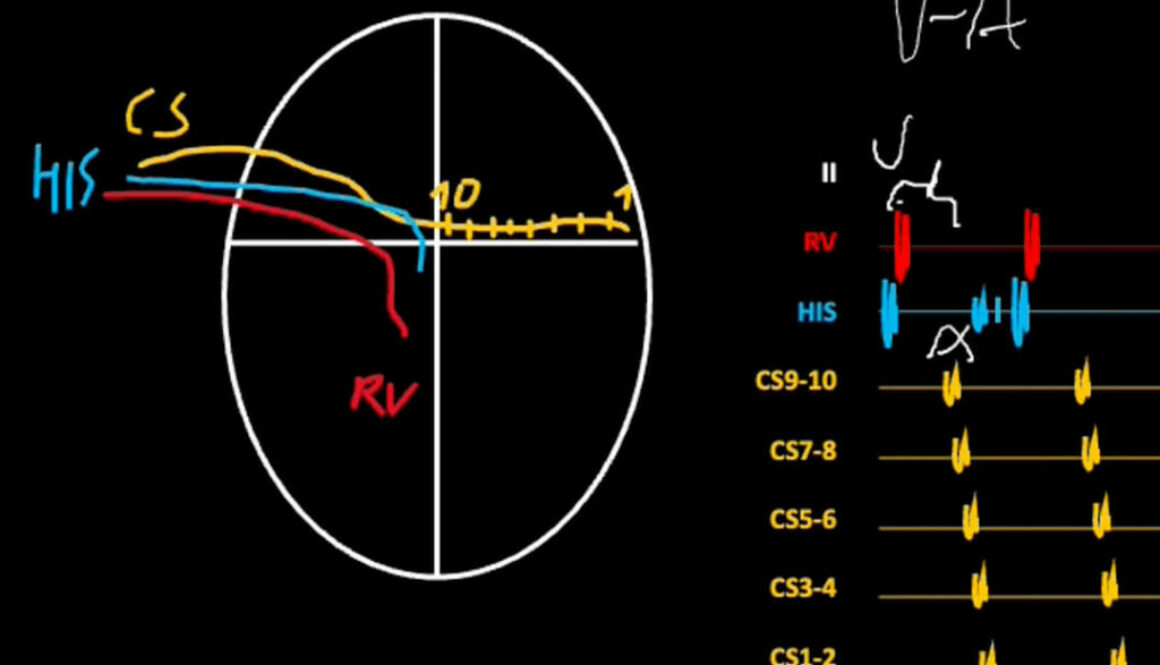
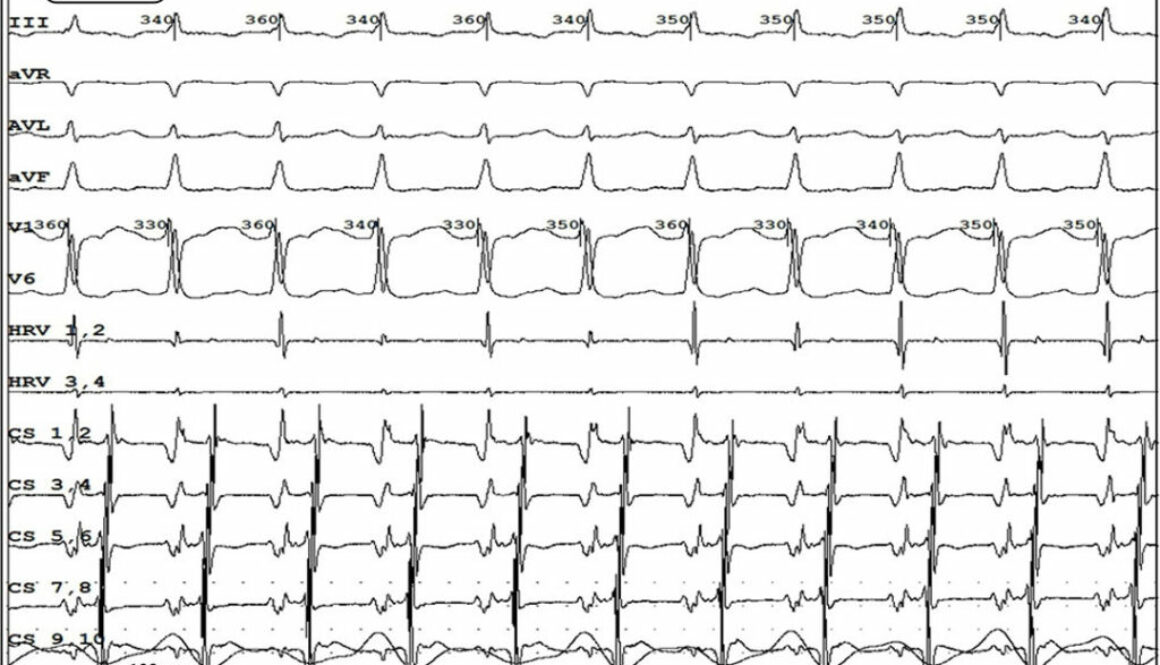
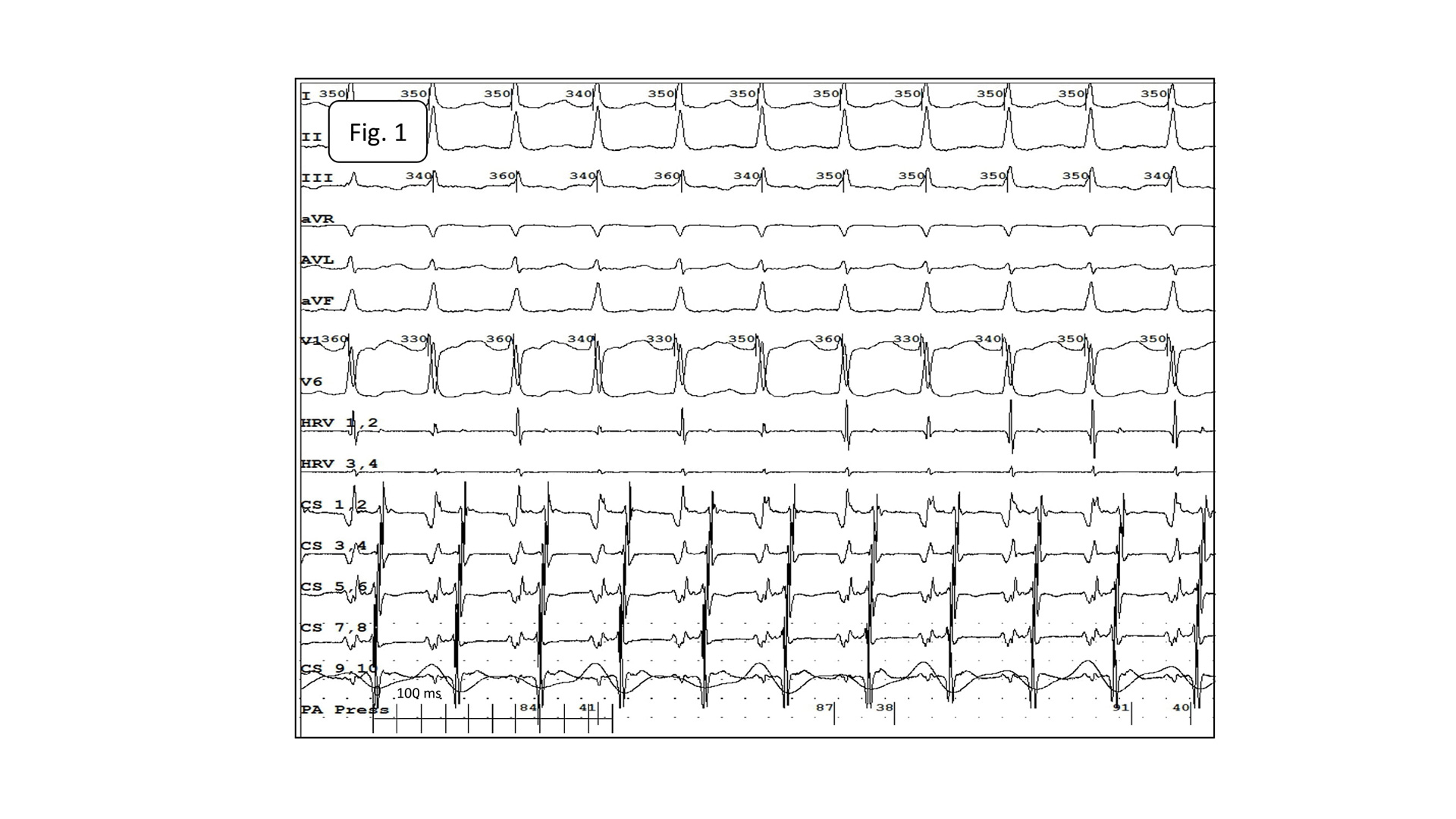
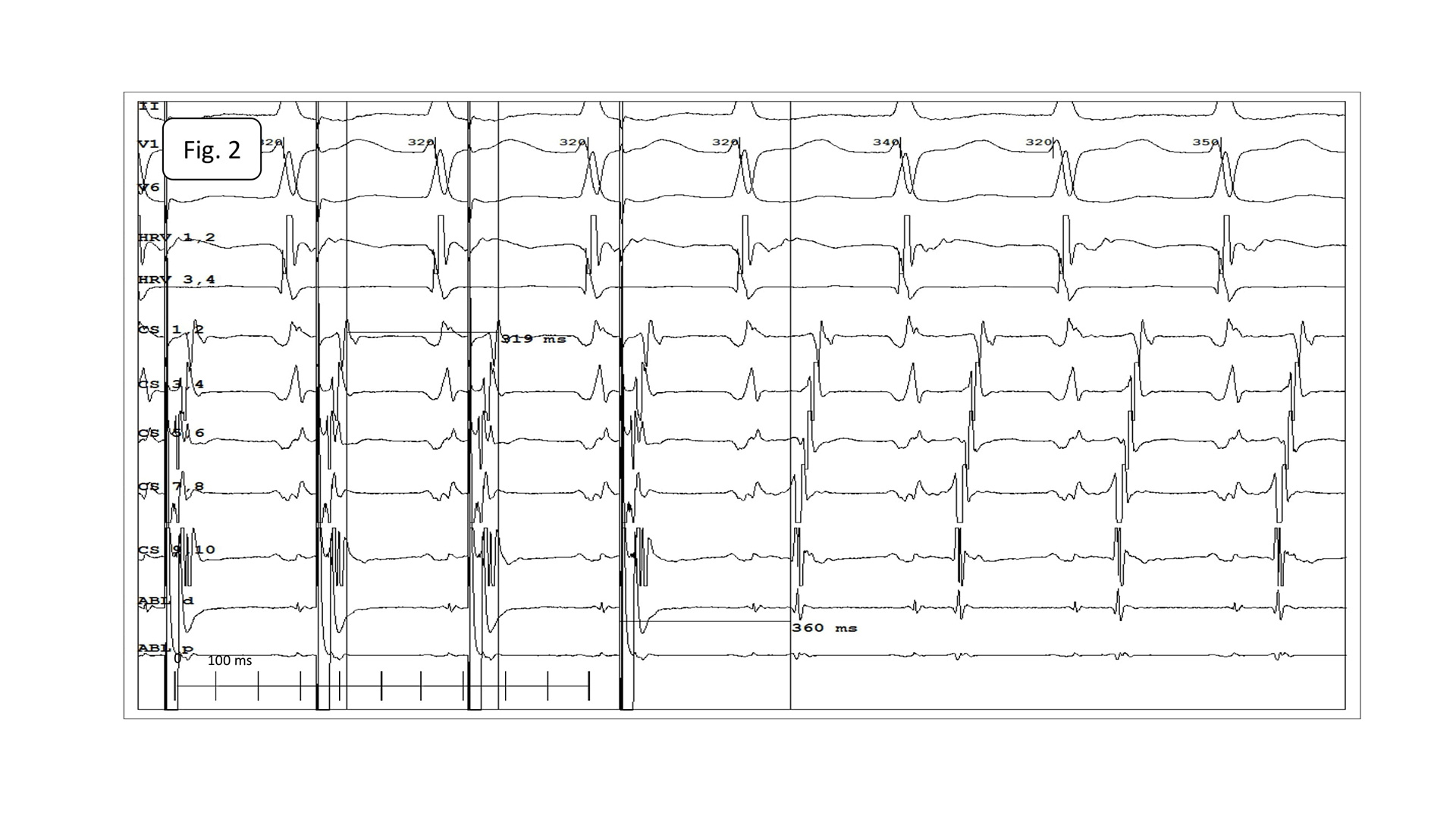
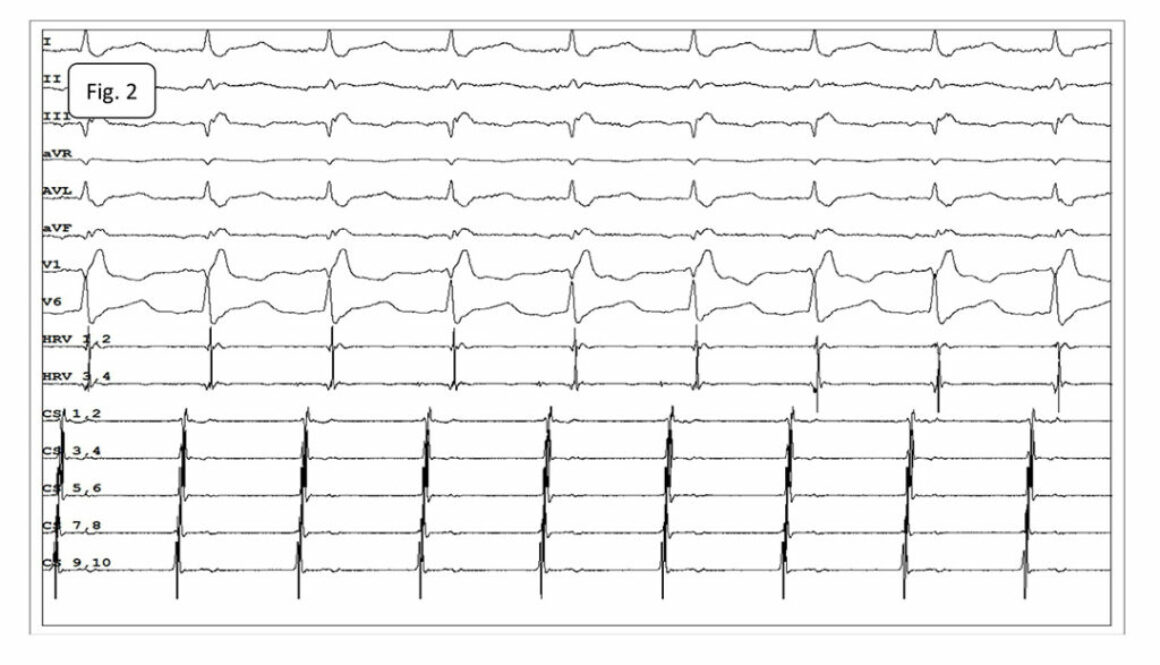
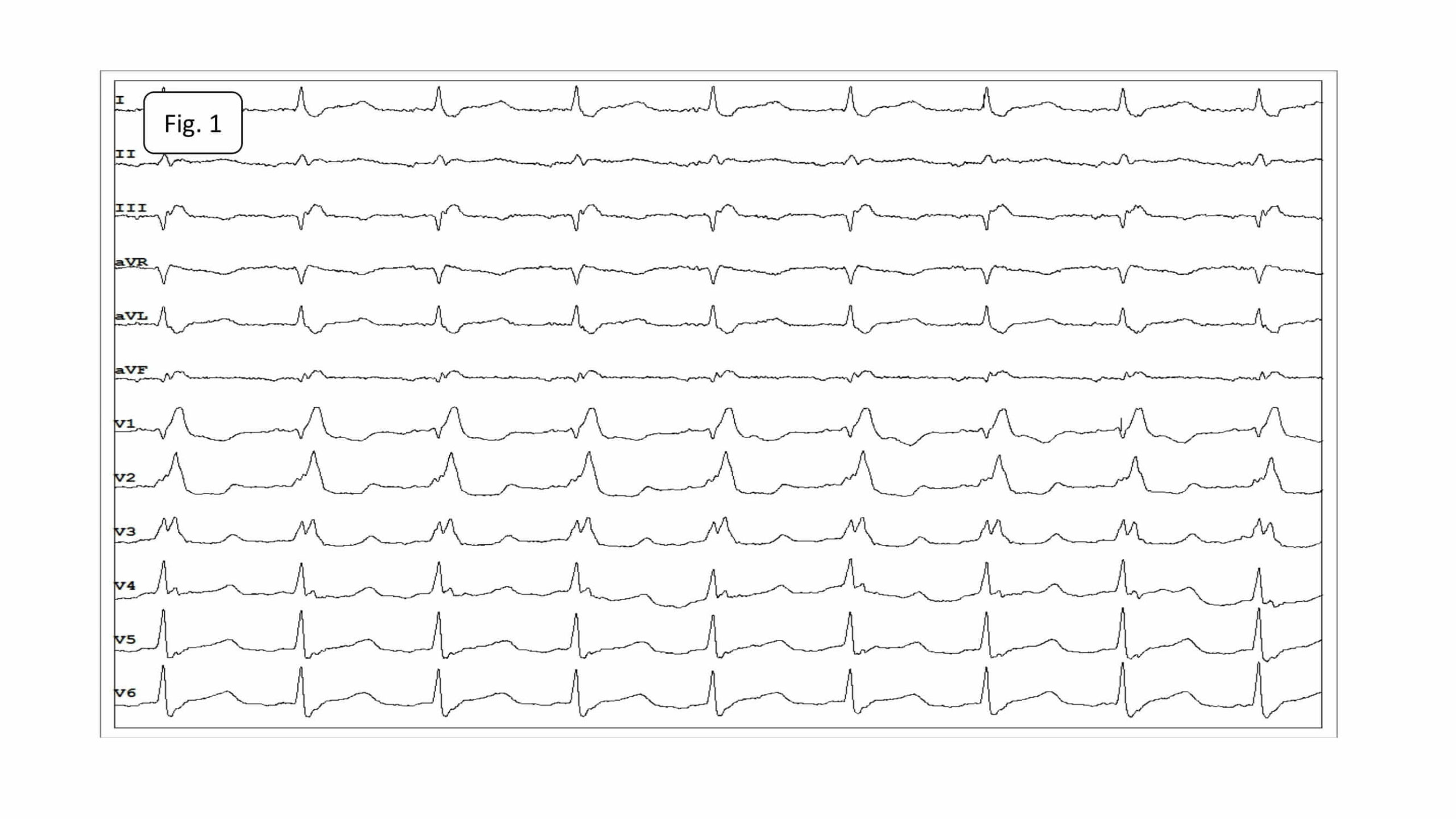
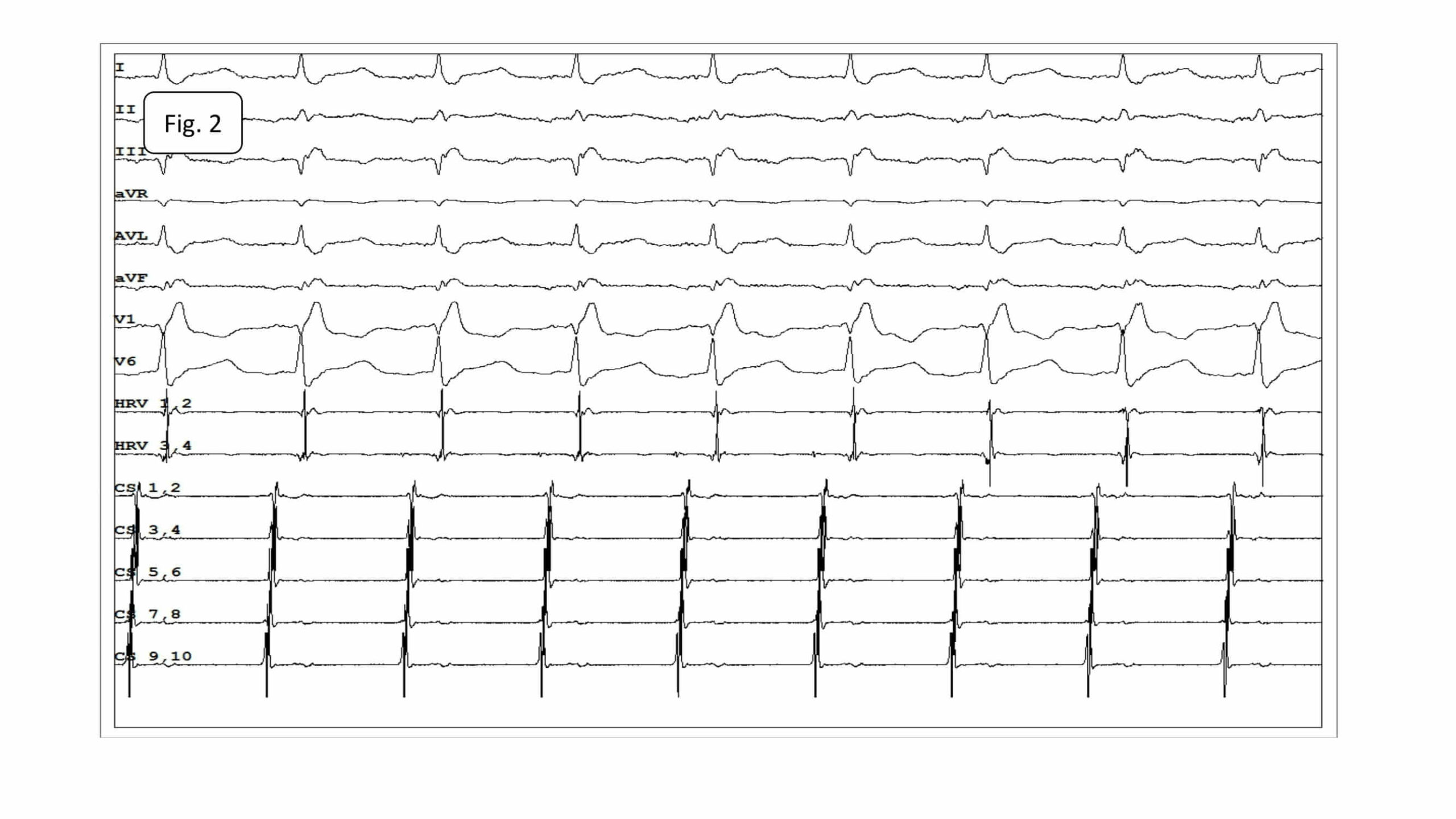
The baseline 12-lead electrocardiogram showed AT with the cycle length of 230-270ms and conduction to the ventricles at a 2:1 and 3:2 ratio. RBBB (Fig.1).
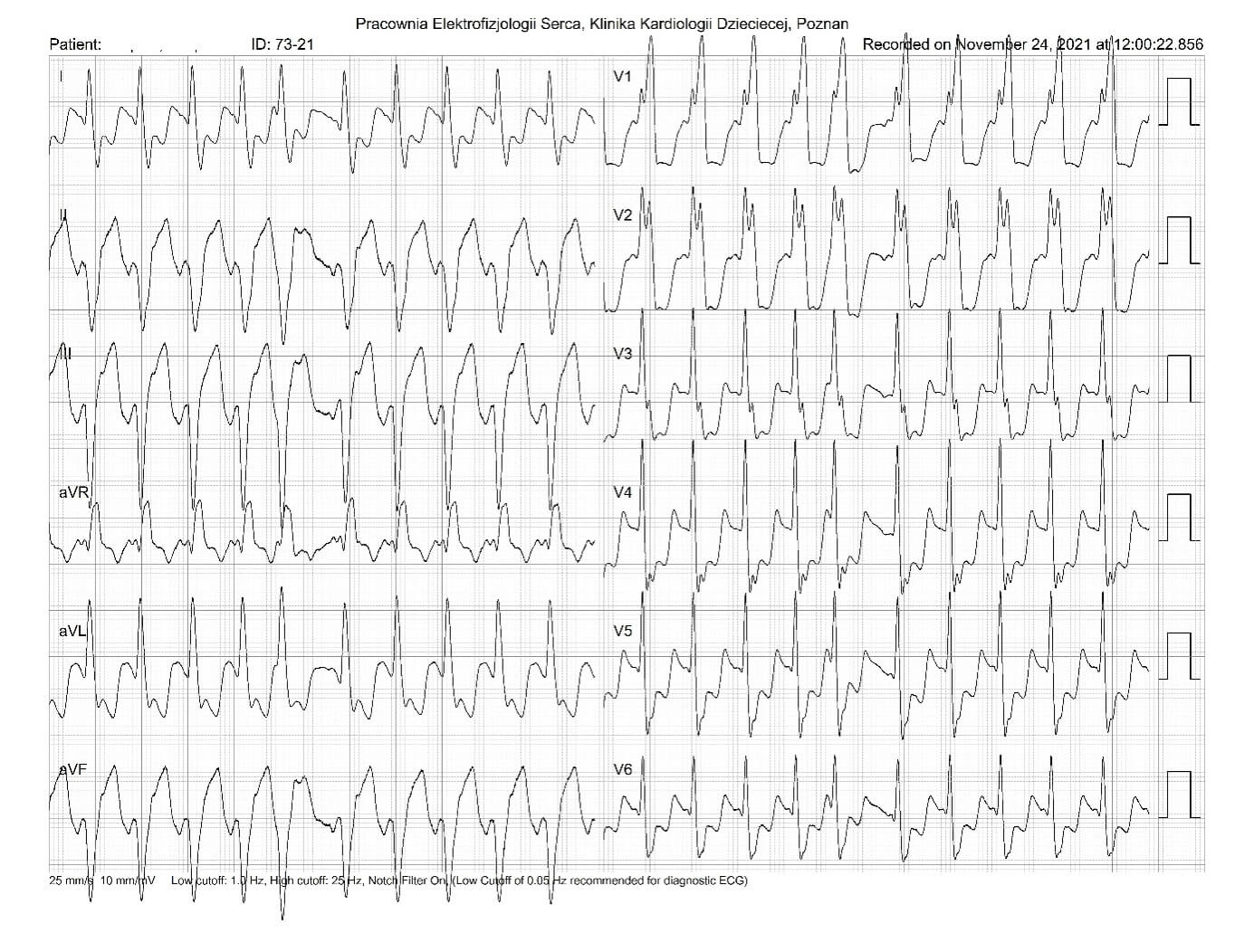
Fig.1. Atrial tachycardia conducted to the ventricles at a 2:1 ratio. RBBB. P-waves of tachycardia best visible in leads: II, III and aVF.
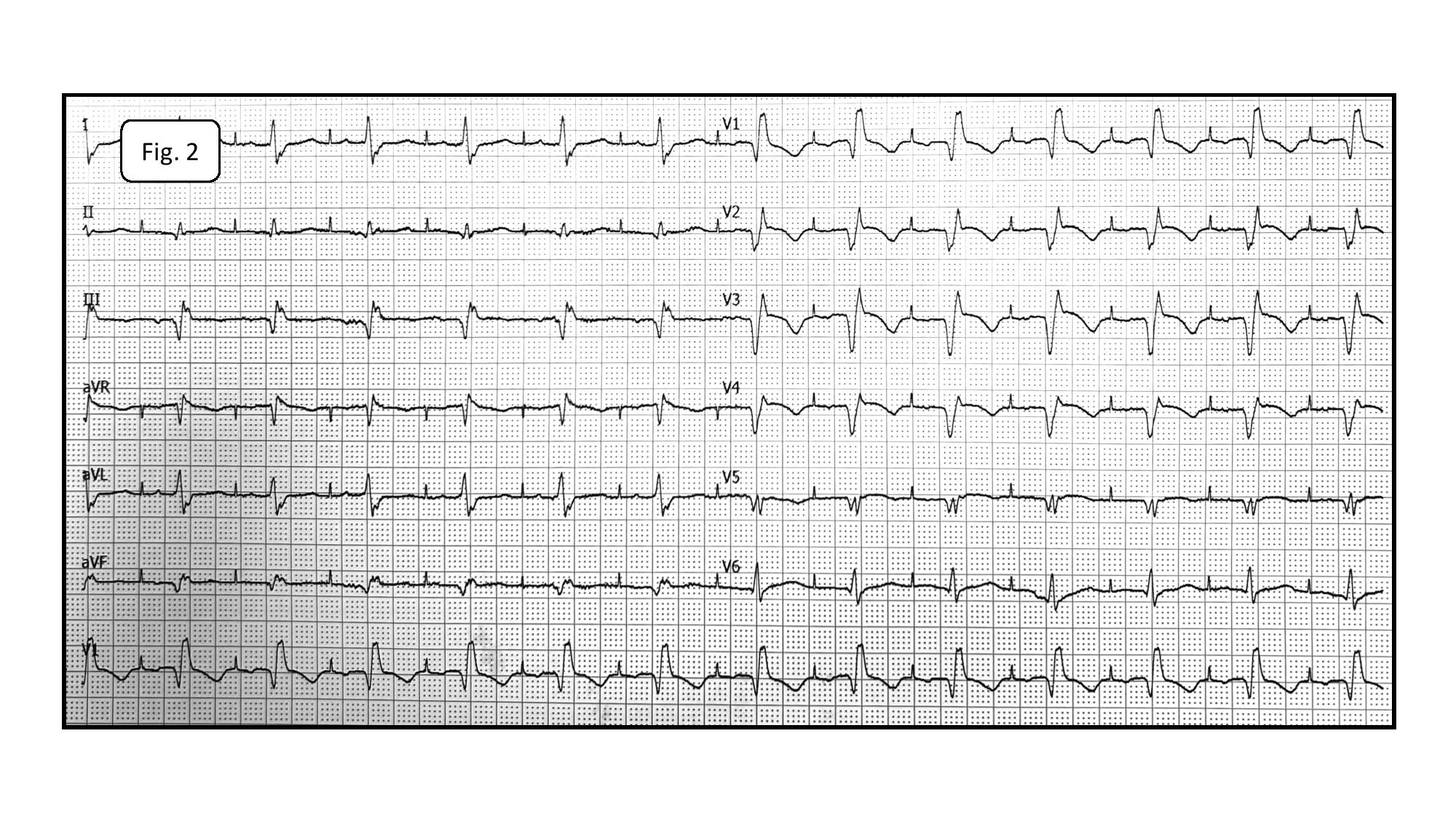
Bilateral femoral vein cannulation was performed with Terumo 4F sheaths as planned, but it was impossible to introduce diagnostic electrodes. Angiography of femoral veins showed iliac veins obstruction with a well-developed collateral circulation (Fig.2). The diameter of those vessels was so narrow that no attempts to introduce hydrophilic leader or microcatheter were made (the technique was performed in certain cases, but almost always operators refrained from placing multiple electrodes in abnormal vessels localized in retroperitoneal space).
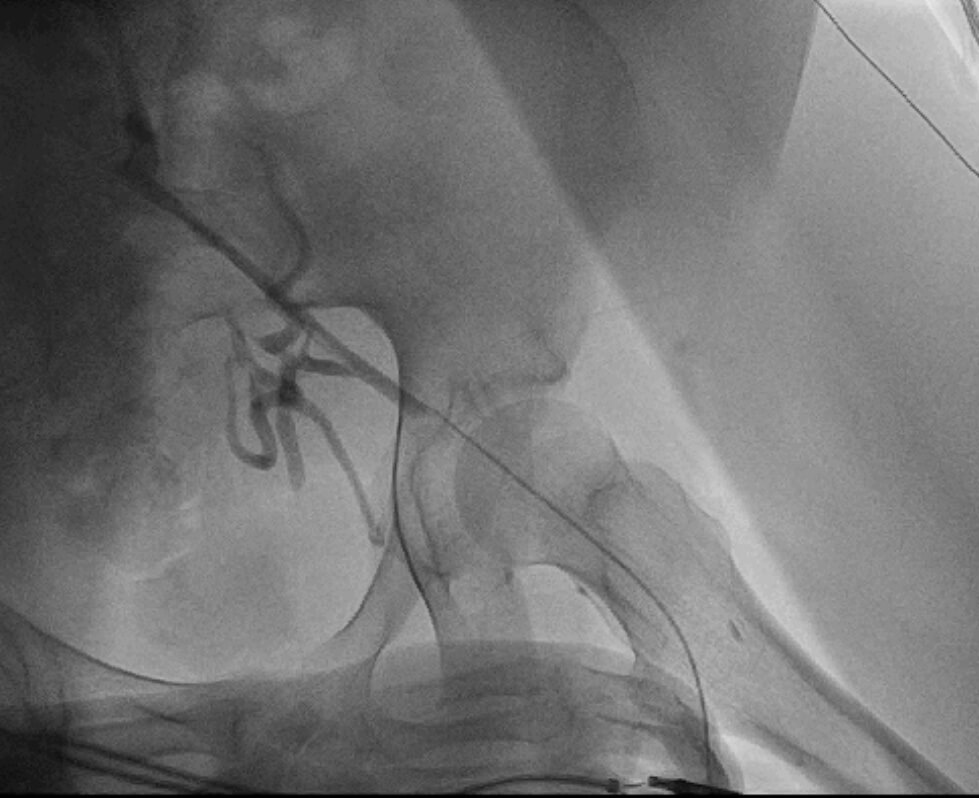
Fig. 2. Angiography of left femoral vein drainage. Contrast was administered via microcatheter introduced along the 0.014’’ angioplasty guidewire (Whisper ES).
As it was impossible to access the heart via inferior vena cava, decision was made to perform 2 internal jugular vein cannulations. Cannulations of the internal jugular vein and the venous angle is performed sometimes in similar situations, depending on weight of the child and anatomical arrangement. Subclavian vein cannulation may result in a additional curvature limiting manipulation of mapping electrodes and of ablation electrode. Short vascular sheaths were chosen for the cannulation to avoid introduction of the equipment into the right atrium. In smaller pediatric patients trimming of the sheaths is even required to achieve this goal.
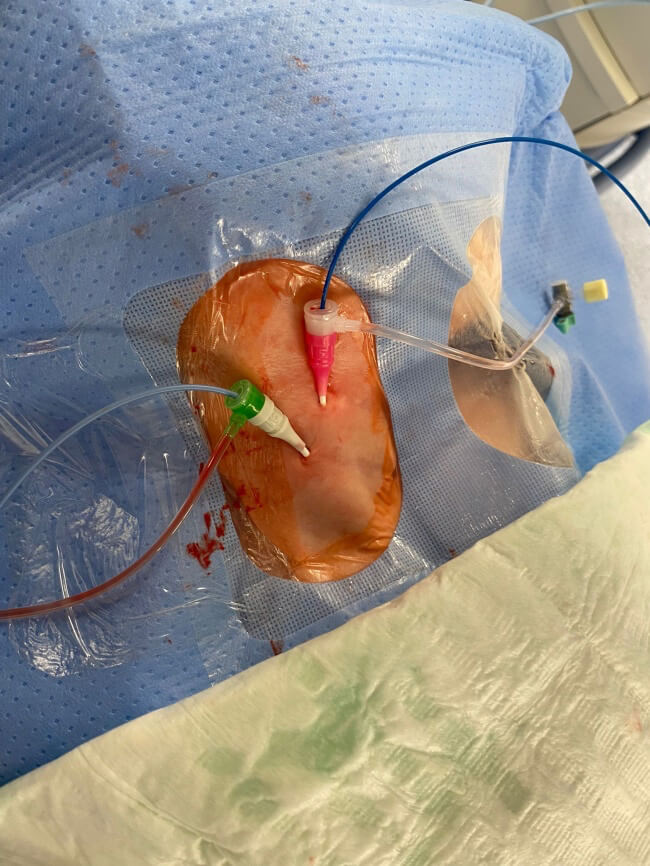
Fig. 3. Ultrasound-guided internal jugular vein cannulation with Doppler imaging. In the first step, 4 and 5F sheaths are introduced to perform electrophysiological study. The proximal sheath is then replaced with 7-8F sheath depending on the electrodes planned to be used subsequently. In younger children attention should be paid to the position of a guidewire during sheath introduction (it should cross the diaphragm) to avoid atrial or ventricular wall perforation. It may be necessary to trim the sheath in order to prevent it from entering the atrium and limiting mobility of electrodes during mapping and ablation.
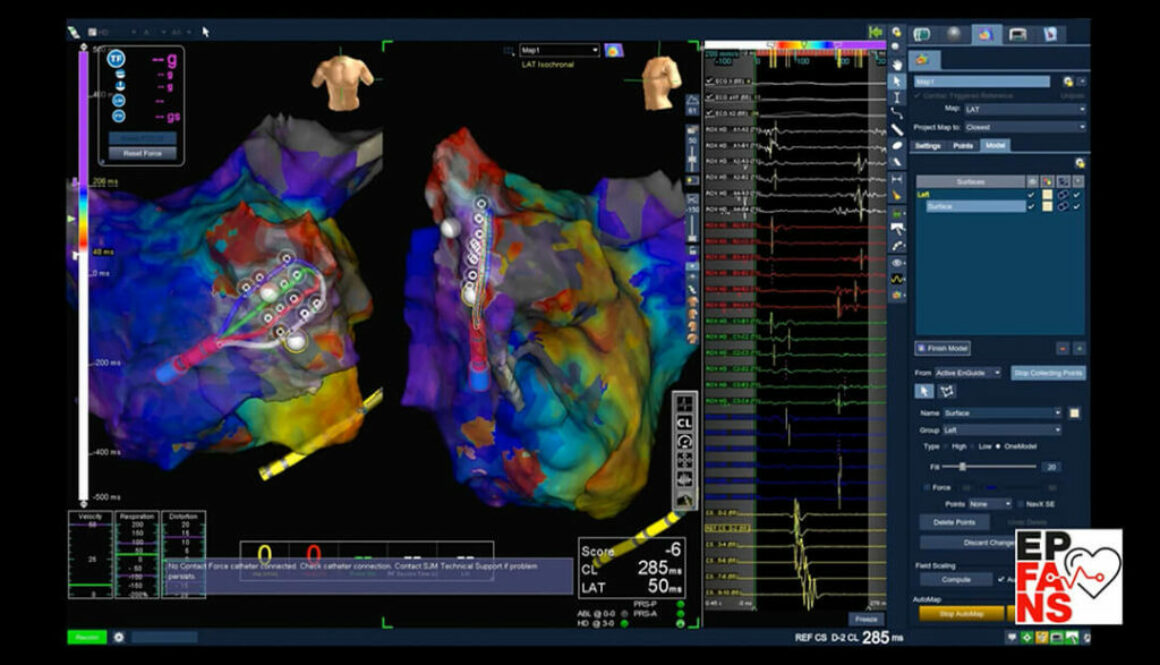
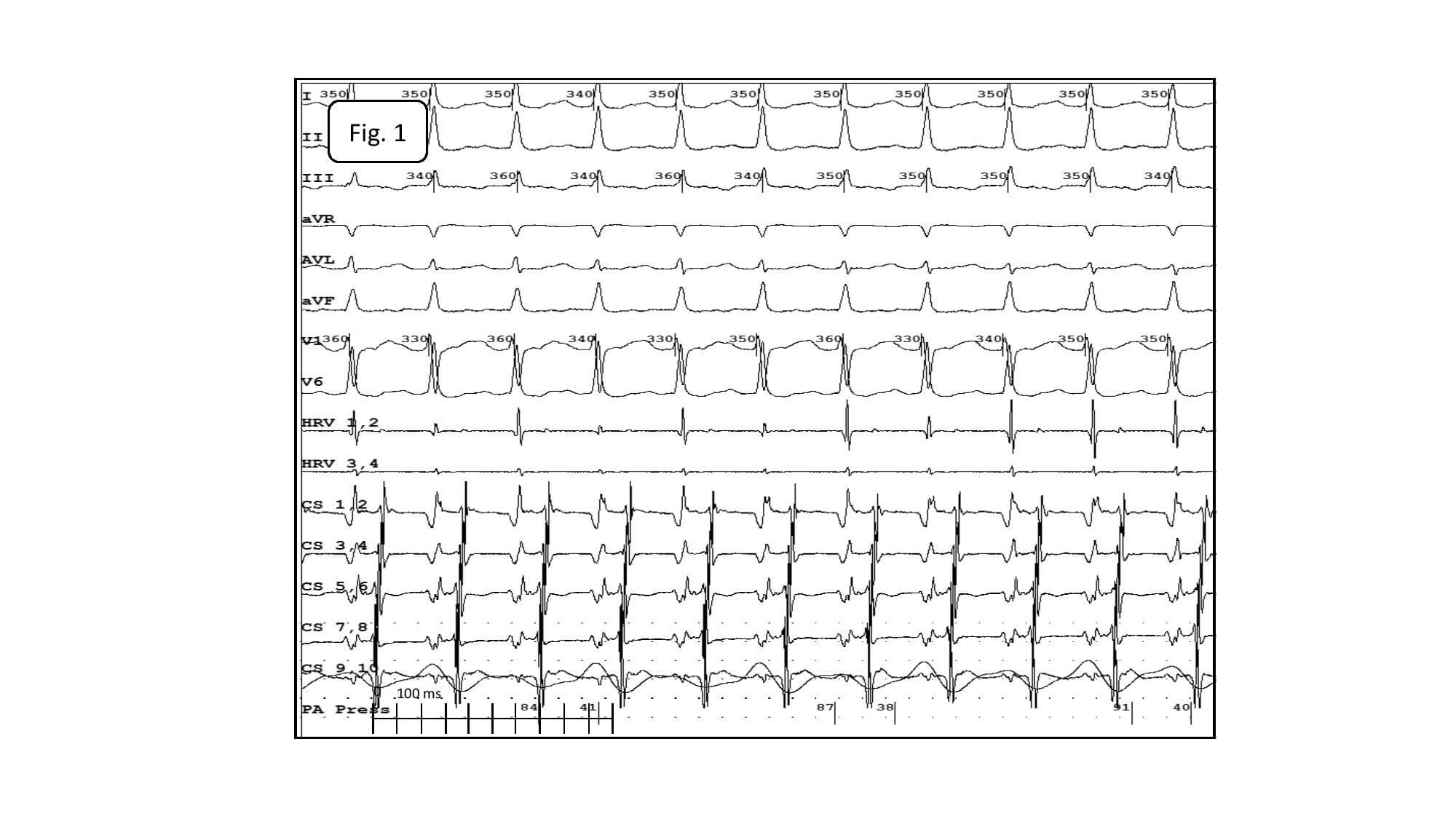
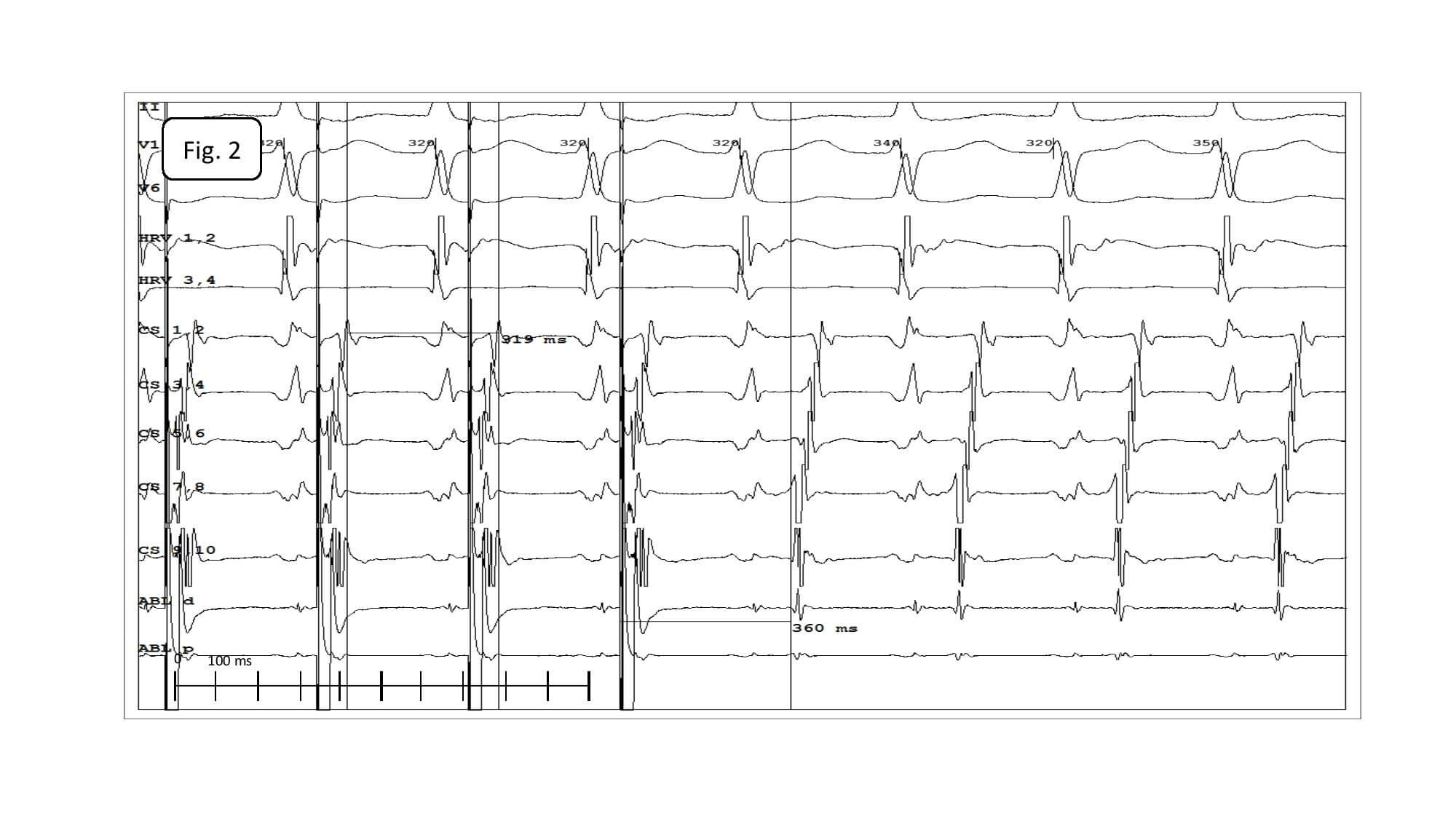
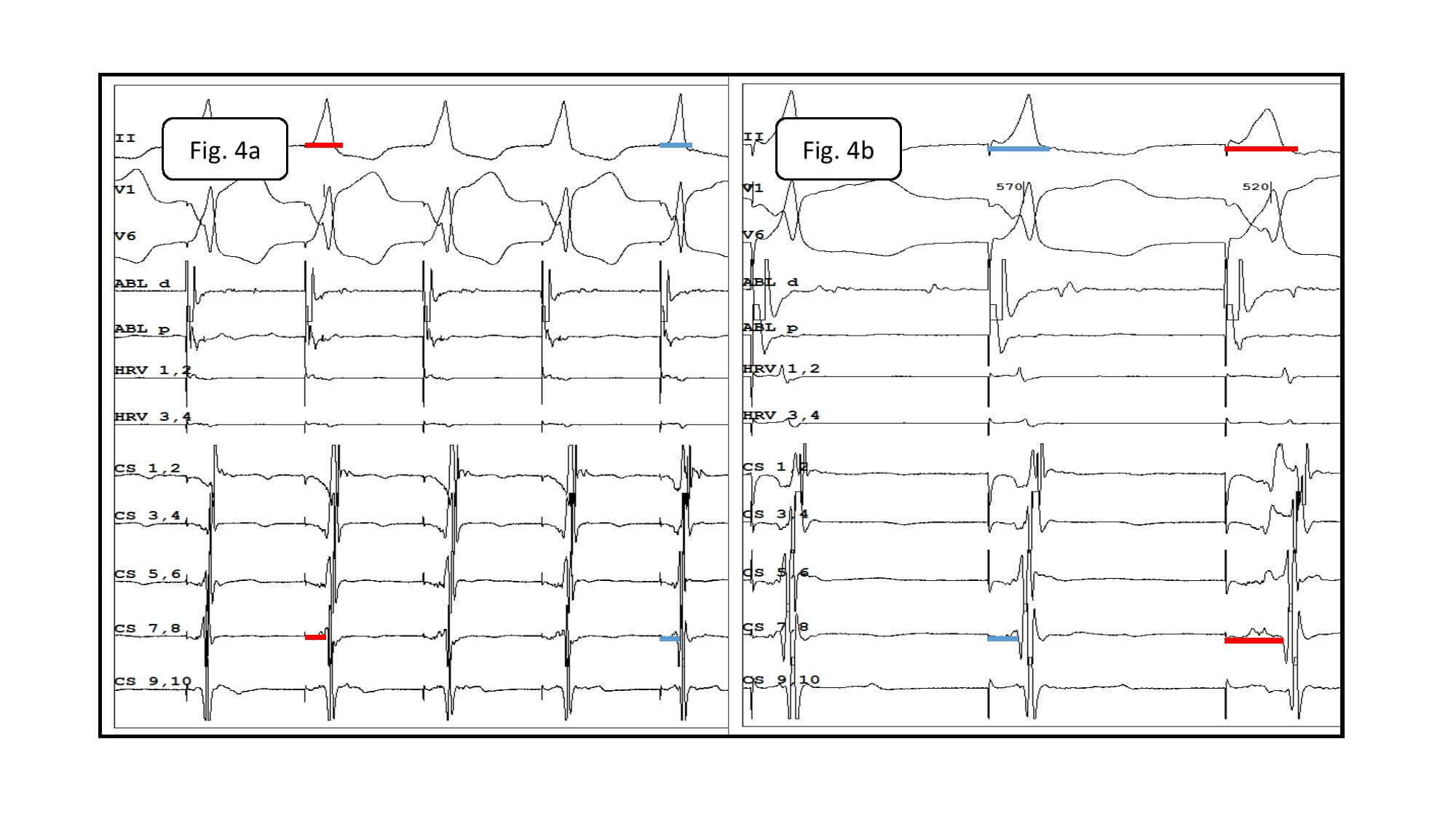
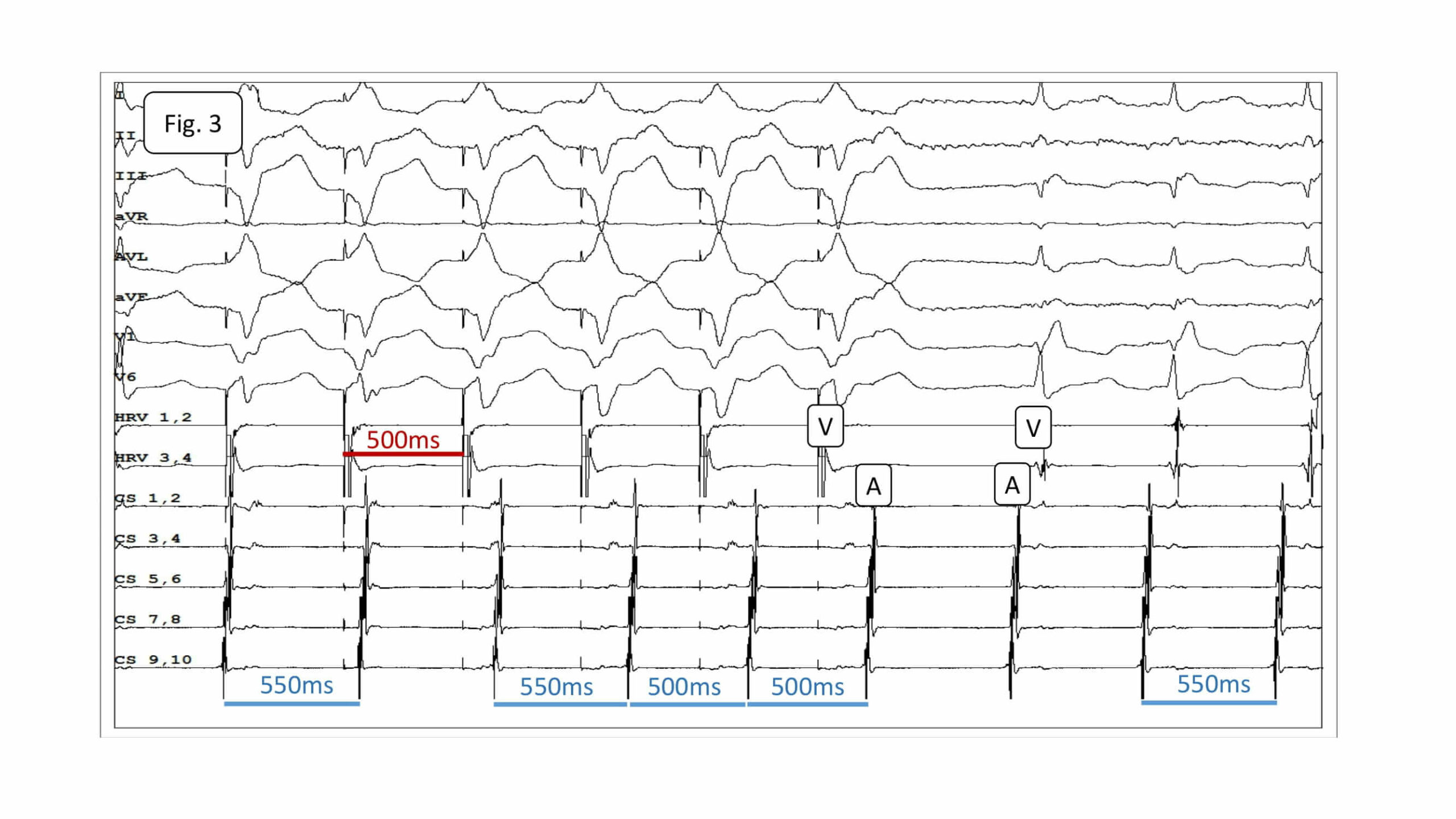
A 5F quadripolar electrode (Supreme, St. Jude Medical) and 5F decapolar steerable electrode (Inquiry, St. Jude Medical) were inserted into the heart and initial assessment of the arrythmia was performed (Fig. 4A). Typical atrial flutter with cycle length of 228ms and localization in the right atrium was confirmed on the basis of the entrainment in the inferior isthmus. Then quadripolar electrode was replaced with the Advisor HD Grid electrode (Abbot Medical). The decapolar electrode was left in the coronary sinus so as to obtain a stable reference. The Advisor HD Grid electrode was chosen for the mapping because of its structure and the concept of activation vectors mapping between respective electrode pairs that not only accelerates the mapping process, but also increases its precision (Fig. 4). Arrangement of the electrodes enables simultaneous bipolar recording of signals along and across the arms. A potential is therefore registered regardless of the direction of depolarization waves propagation. The shape of the Advisor HD Grid electrode allows easy registration of potentials thanks to the stability of its position in the atrium (Fig. 5 and 6). The design of this electrode, free from potentially irritating elements, protects against extrasystoles provocation, so it does not affect significantly the risk of arrhythmia termination during the mapping.
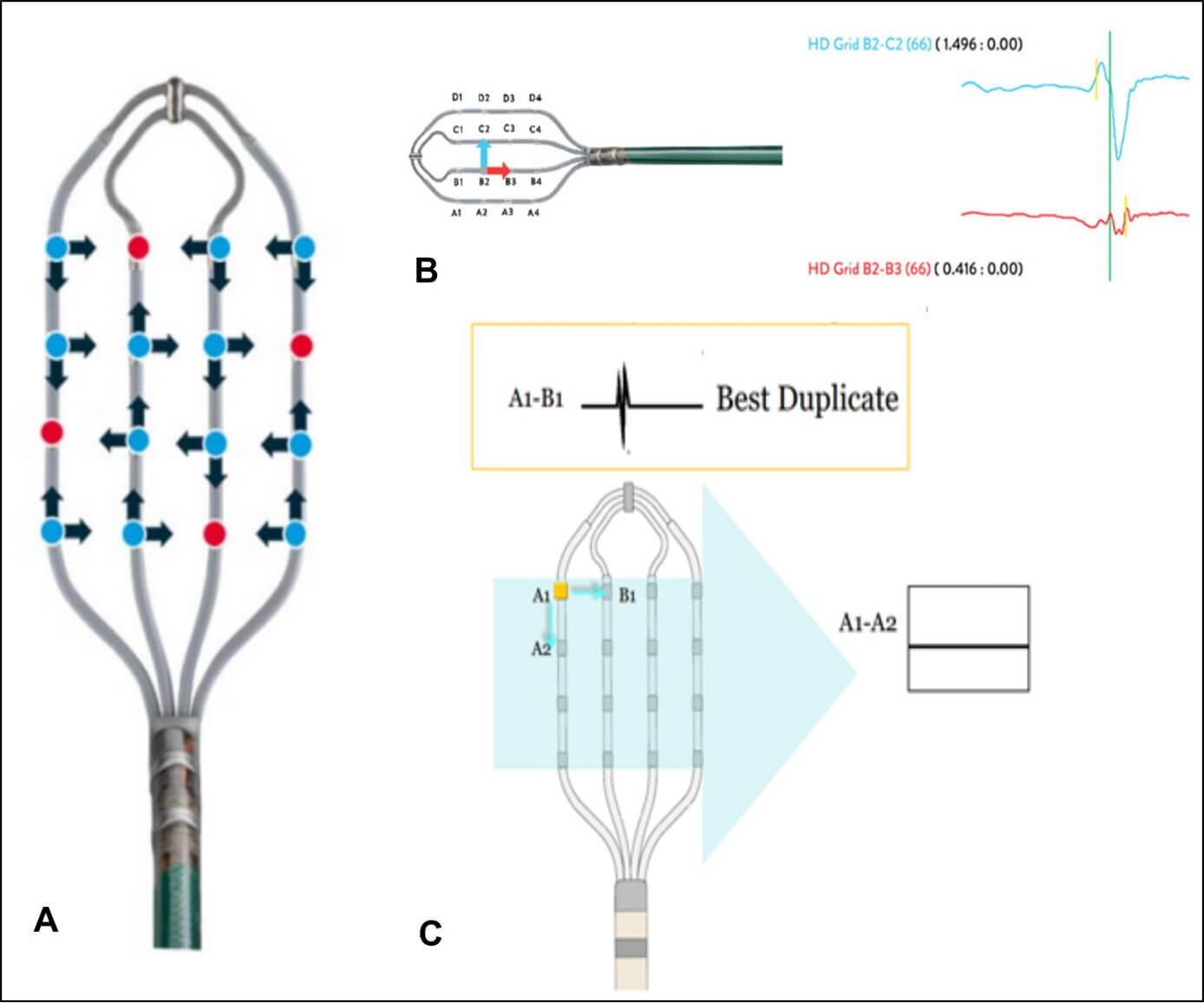
Fig. 4. The structure of the Advisor HD Grid electrode. A. An exemplary arrangement of electrode pairs registering depolarization wave. B and C. Explanation of the potential registration process depending on the direction of depolarization wave propagation in relation to respective electrode pairs.
A:

B:
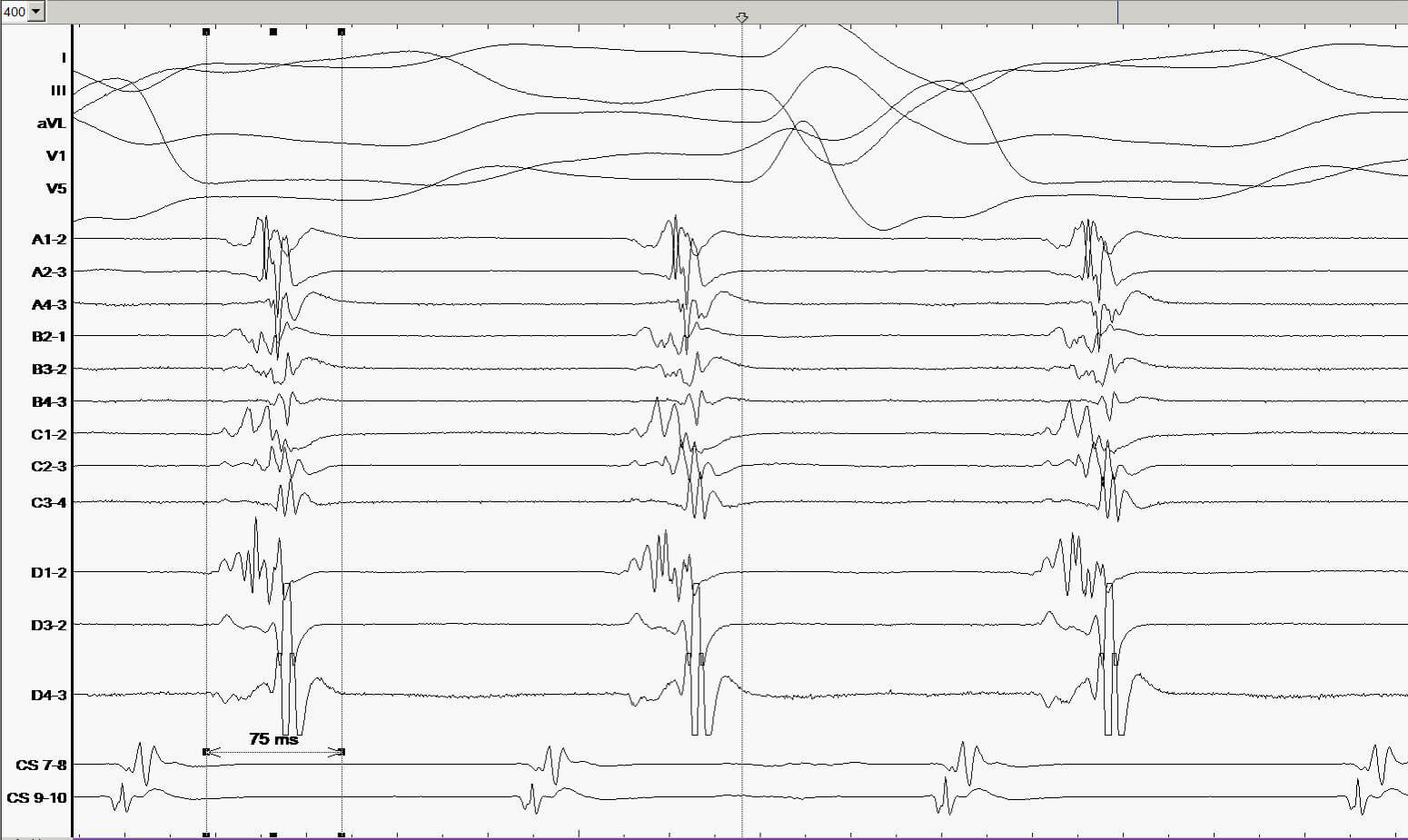
Fig. 5 A. Intracardiac potentials in the electrophysiological study. Tachycardia with cycle length of 228ms with concentric activation on electrode located in coronary sinus. B. Fragmented potentials registered by the Advisor HD Grid electrode from the postoperative scar.
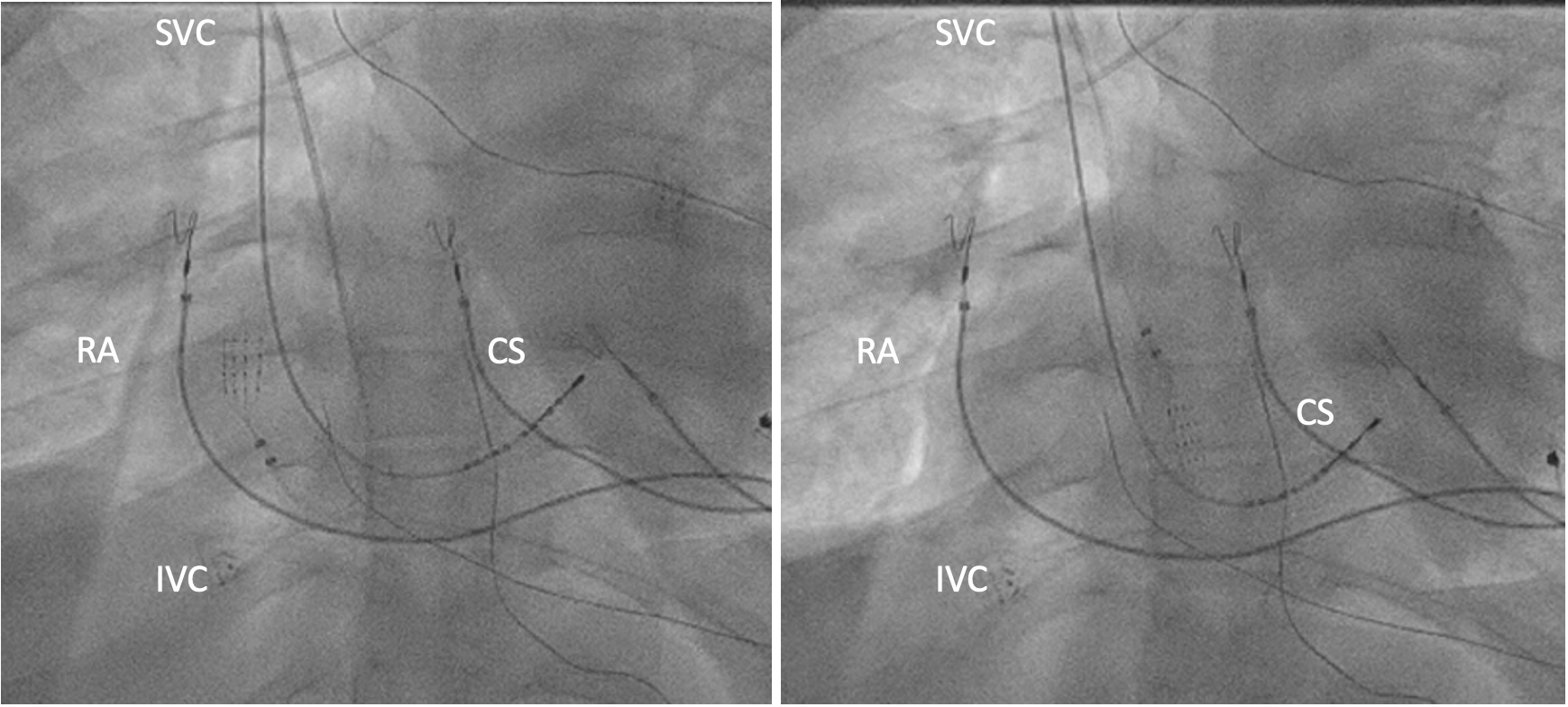
Fig. 6. Mapping of the right atrium with the Advisor HD Grid electrode introduced via internal jugular vein access (SVC – superior vena cava, RA – right atrium, IVC – inferior vena cava, CS – coronary sinus)
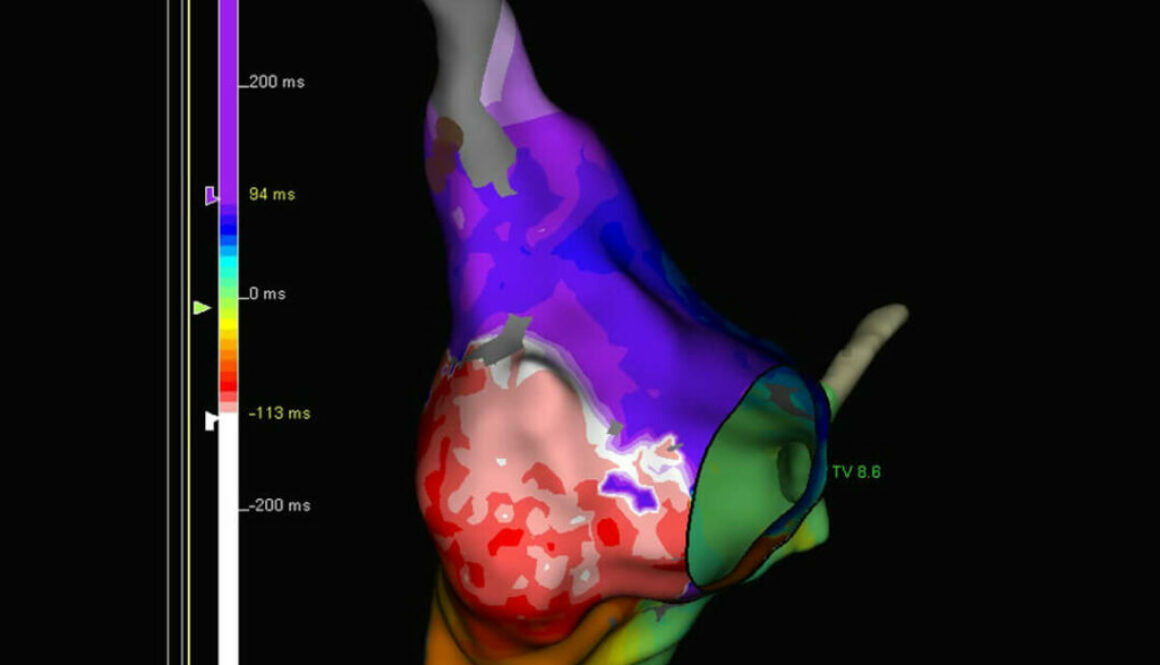
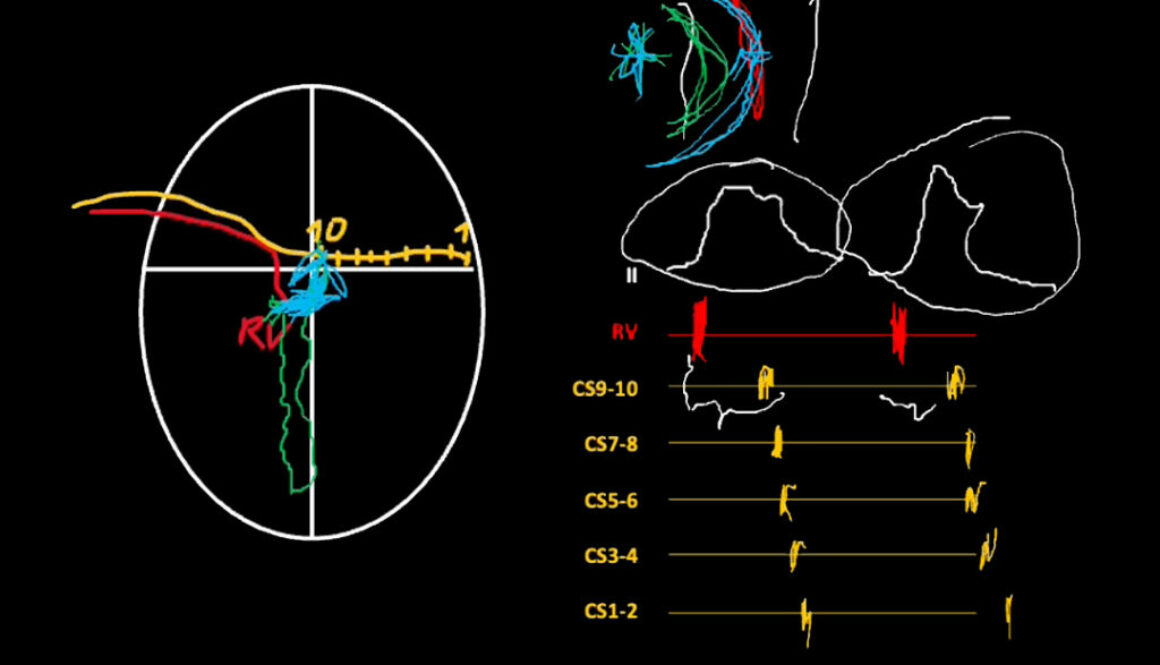
An obtained high-resolution map of the right atrium made during intra-atrial reentrant tachycardia (IART) showed a small scar in the postero-lateral area of the right atrium (Fig. 7). A slow conduction zone was also localized between the scar and the tricuspid annulus on a propagation map (Fig. 8). An entrainment was performed in that spot, each time leading to arrythmia termination. However, IART was then induced by rapid atrial stimulation.
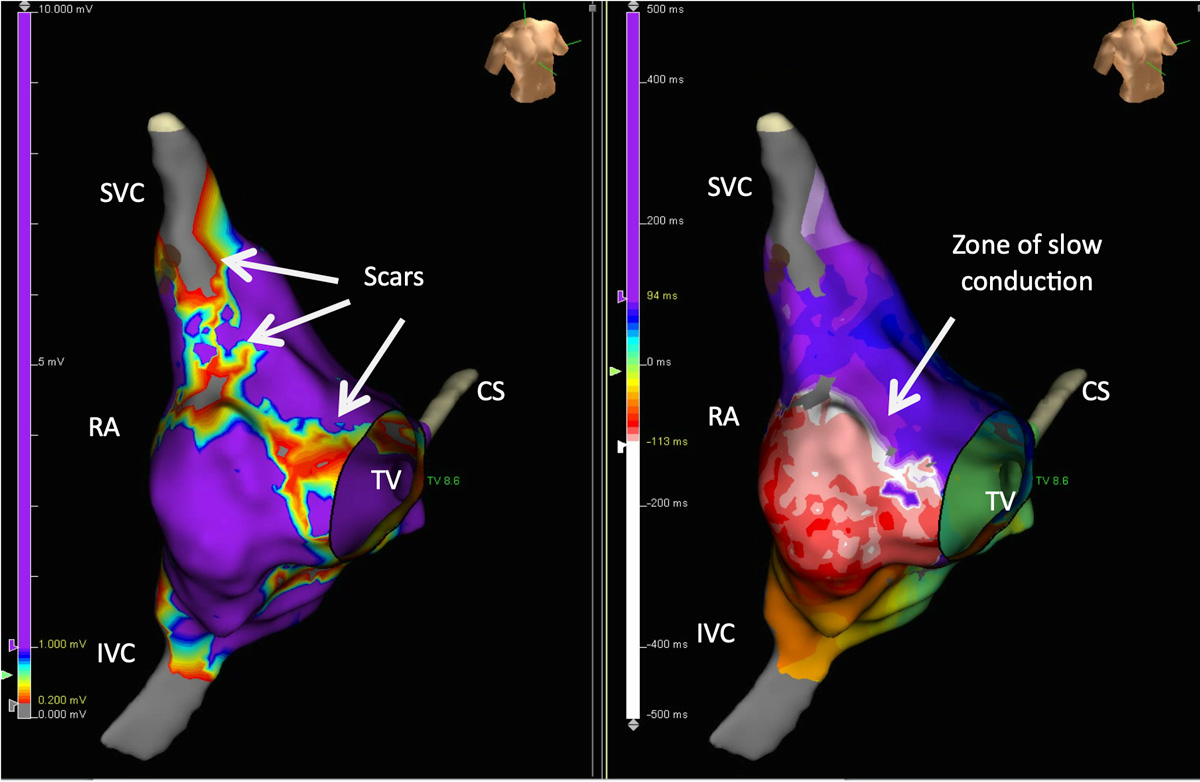
Fig. 7. Potential map (A) and propagation map (B) made during tachycardia. A low-voltage zone was localized between the scar in the postero-lateral area of the right atrium and the scar localized near the lateral aspect of the tricuspid annulus. This is the zone where the propagation of signals is slowed down (the isthmus for a macro-reentrant tachycardia). An entrainment in that spot led each time to arrythmia termination. (SVC – superior vena cava, RA – right atrium, IVC – inferior vena cava, CS – coronary sinus, TV – tricuspid valve)
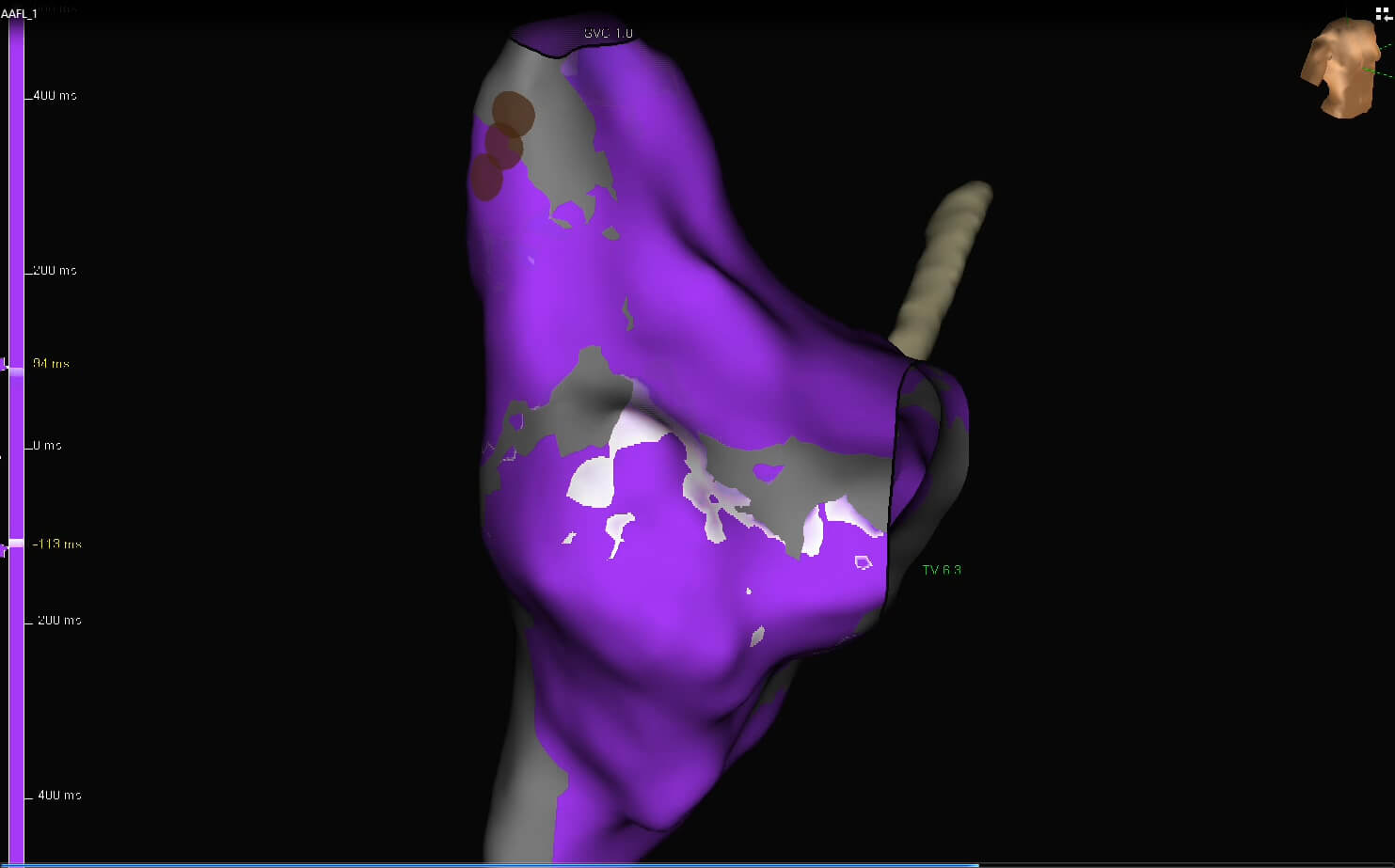
Fig. 8. Propagation map identifying the zone with significantly slowed down conduction between the scar in the postero-lateral area of the right atrium and the tricuspid annulus. In postoperative atrial tachycardias this zone is most commonly responsible for macro-reentry in the right atrium.
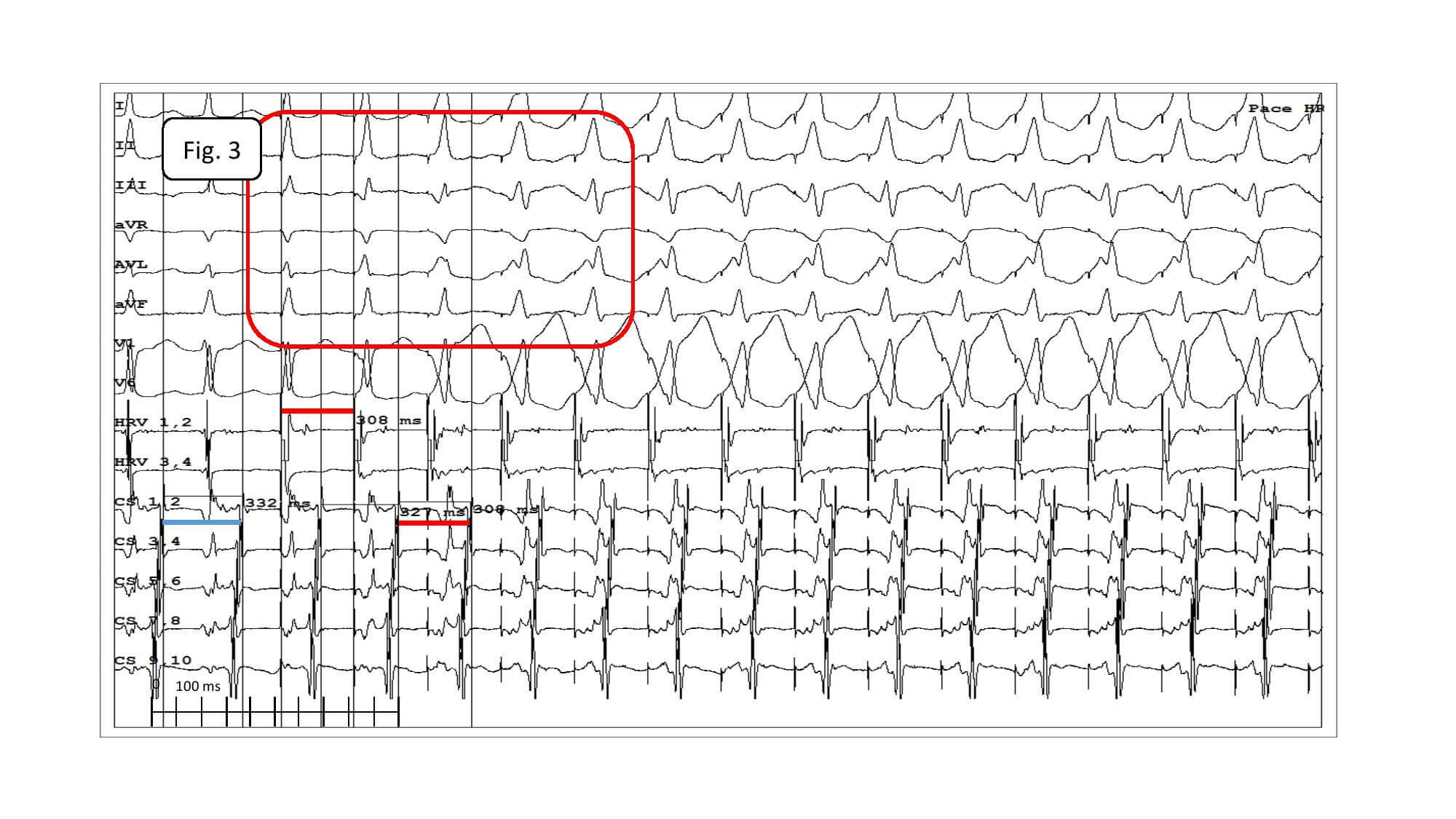
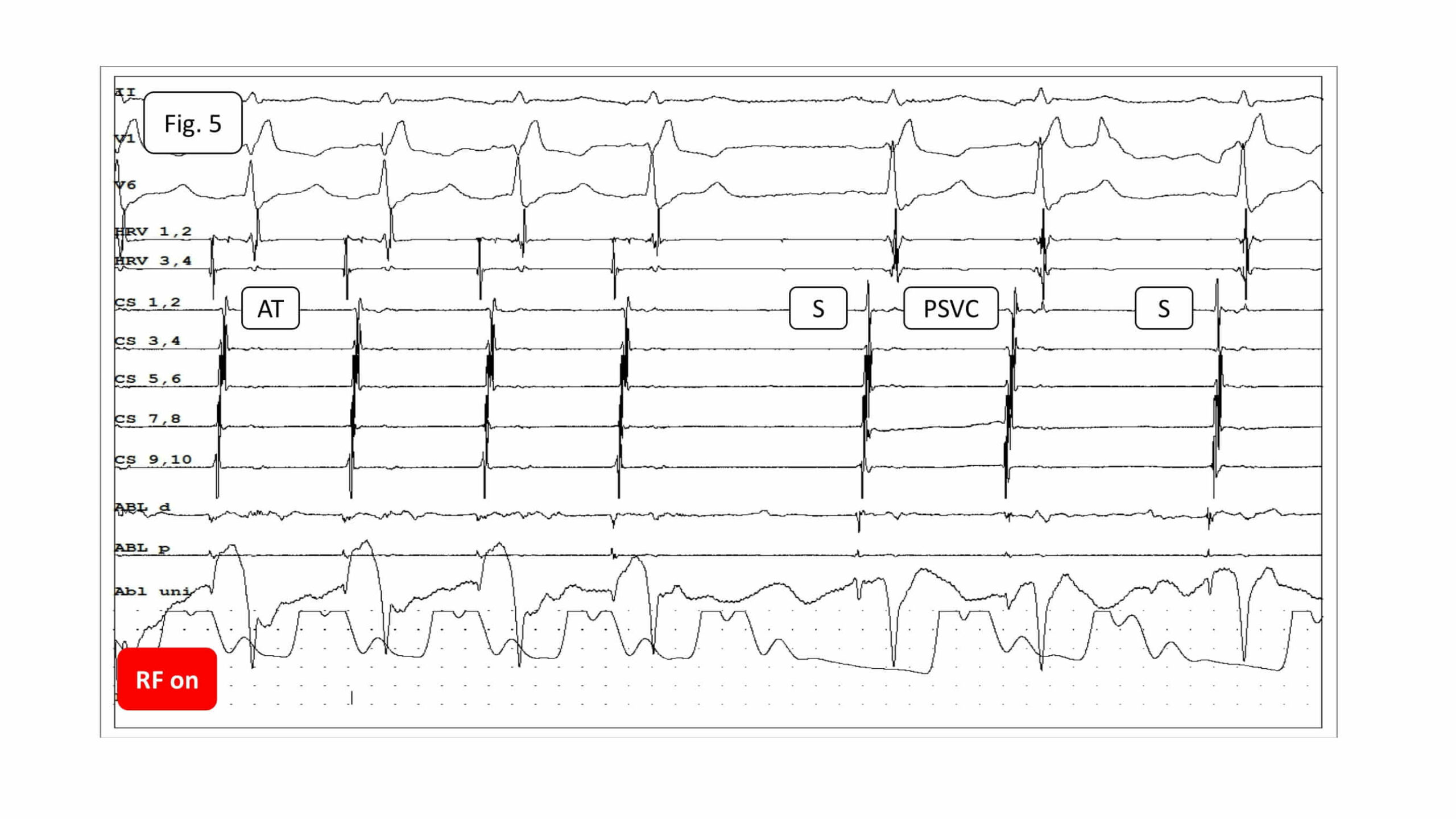
Having analysed the potential and propagation maps, a linear ablation with a cooled electrode between the scar and the tricuspid annulus was planned. An RF application started in the area of fragmented potentials (Fig. 9). The arrhythmia was terminated during that first application (40th second). Nevertheless, a complete application line was made between previously identified scar zones (Fig. 10.). Due to common concomitance of typical, cavotricuspid isthmus(CTI)-dependent atrial flutter a linear application was made in the CTI achieving a bidirectional block. Then a remap was created during a stimulation of coronary sinus. The Advisor HD Grid electrode was used to confirm the sealing of the application lines. Additional applications were performed to extend the line on the lateral wall towards the scar (total application time – 696s).
After the procedure the EPS was performed and revealed ortodromic and retrograde conduction via AV node with decrement. ERP AVN ante 260 ms. On CS stimulation 85/min.: PQ 80 ms. QRS 155 ms (RBBB). Wenckebach point 178/min. Programmed and rapid atrial stimulation induced no arrythmia.
Patient was discharged home on the 3rd day after procedure. During a Holter monitoring on the day of ablation and after 6 months no tachycardia recurrences were registered (some isolated or grouped extrasystoles only).
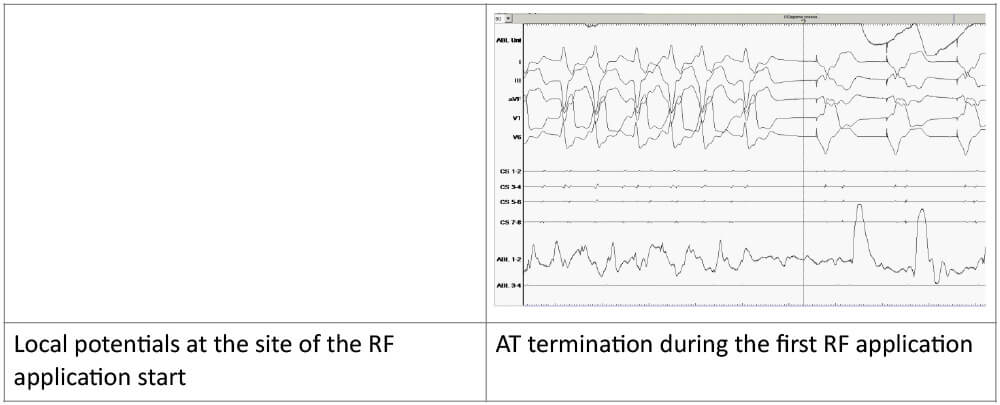
Ryc. 9. Local fragmented potentials at the site of application start and the moment of AT termination. Due to sinus arrest, ventricular stimulation is activated. (AT – atrial tachycardia, RF – radiofrequency)
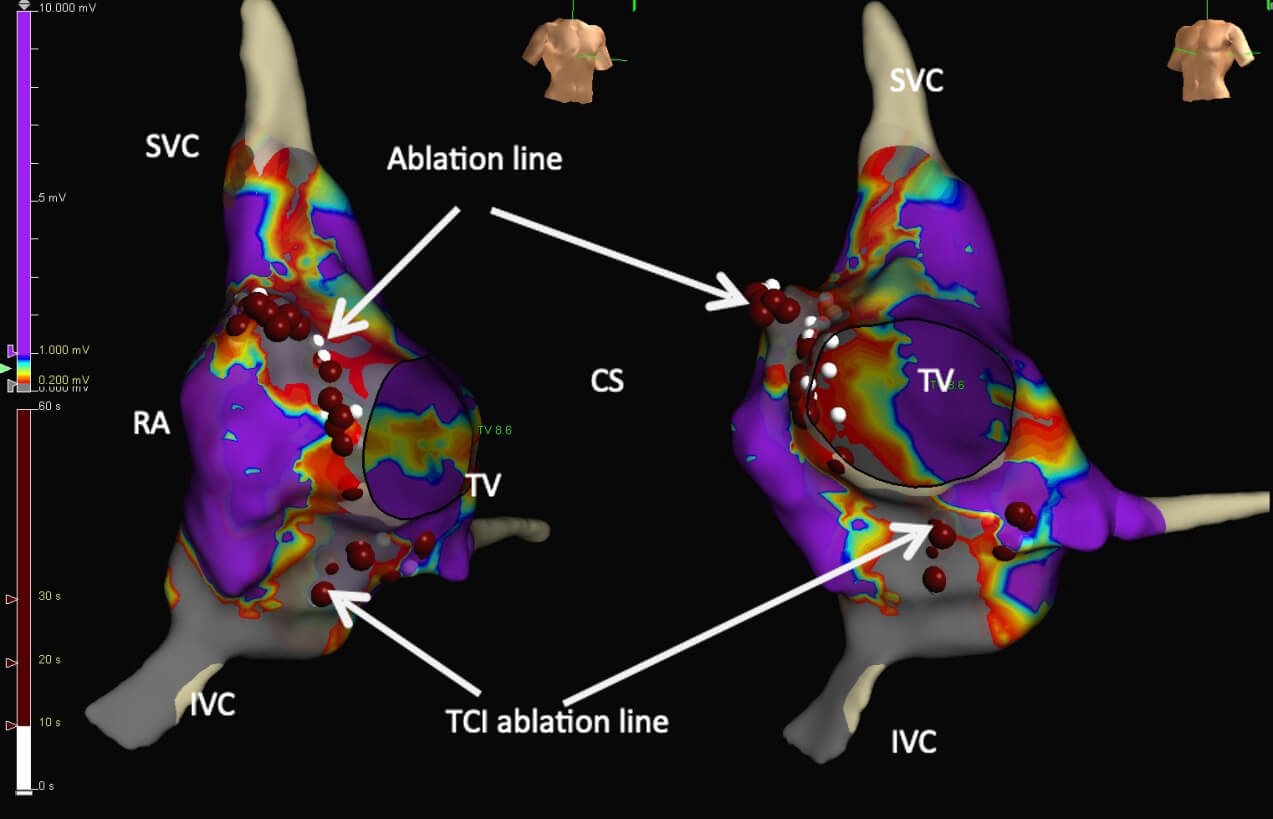
Fig. 10. Linear application between the scar in the postero-lateral area of the right atrium and the tricuspid annulus, and another linear application between the tricuspid annulus and the IVC.
(SVC – superior vena cava, RA – right atrium, IVC – inferior vena cava, CS – coronary sinus, TV -tricuspid valve, TCI – tricuspid caval isthmus)